Scale-up of adherent Vero cells grown on Cytodex™ microcarriers using single use bioprocessing equipment
Posted on November 4, 2014
In cell culture-based vaccine production, scale-up of adherent cells is challenging. This study shows a process for scaling up adherent Vero cells from static cell factories to influenza production at 50 L scale using WAVE Bioreactor™ systems and ReadyToProcess™ singleuse bioprocessing equipment. Vero cells were grown to high cell density on Cytodex microcarriers in 10 L working volume. The cells were detached with trypsin and used to seed a 50 L production culture with the same microcarrier concentration. The cells were allowed to reattach and grow on the new microcarriers in a larger Cellbag™ bioreactor chamber. Cells were subsequently infected with influenza virus. The results show a repeatable scaleup procedure. The cell growth was similar in the 10 L seed cultures as in the 50 L production cultures in three consecutive experiments, with an obtained hemagglutinin (HA) concentration of approximately 12 μg/mL and a virus titer of 109 virus particles/mL.
Introduction
Compared to an egg-based vaccine production, cell culturebased vaccine production is seen as an alternative way for a more rapid response to pandemic challenges. Traditionally, cell-based vaccine production is performed in stainless steel bioreactors requiring extensive cleaning procedures. With disposable bioreactor systems without the need for cleaning and validation, the start-up time for GMP manufacturing can be significantly reduced (1).
Adherent cells, such as Vero cells, are almost exclusively used for human cell-based vaccine production. Vero cells are susceptible to a broad range of viruses and are widely used for the production of viral vaccines like polio, rabies, or enterovirus (2, 3). Recently, influenza vaccine was successfully produced at industrial scale using Vero cells (4). Vero cells can only proliferate when provided with a suitable surface. The scale-up of such cell cultures is challenging because of the required detachment and reattachment of cells during passaging. The aim of this study is to present a process for scaling up adherent Vero cells for influenza propagation. The cultures were run at a working volume of 50 L using WAVE Bioreactor system and ReadyToProcess single-use equipment (Fig 1). When scaling up to 50 L from a 10 L bioreactor culture, the cells were detached by trypsinization and subsequently used to seed new Cytodex microcarriers of the same concentration in the larger Cellbag bioreactor. The cells reattached and grew on the new microcarriers and were subsequently infected with influenza virus. Our results show that the scale-up procedure using single-use WAVE Bioreactor systems is a fast and convenient option for virus production.
Materials and methods
Cell line and cultivation media
Vero cells were obtained from ATCC (No. CCL81, ATCCLGC Standards, Teddington, Middlesex TW11 0LY, UK). The cultivation medium used was Dulbecco’s modified Eagle medium (DMEM)/Ham’s F12 (GE Healthcare Life Sciences) supplemented with glucose (1 g/L), glutamine (0.9 g/L), β -cyclodextrin (0.1 g/L) (Sigma-Aldrich Co., St. Louis, MO, USA), and soy peptone (2 g/L) (Kerry, Norwich, NY, USA). For bioreactor cultivations, Pluronic™ F-68 was added (2 g/L) (Sigma-Aldrich Co.). For cell propagation, the cultivation medium was supplemented with fetal bovine serum (50 g/L) (GE Healthcare Life Sciences). Inoculum was grown in Nunc™ T-flasks and Cell Factory™ systems (Thermo Fisher Scientific Inc., Waltham, MA, USA). For detachment, cells were washed with PBS-EDTA and incubated with Accutase™ (GE Healthcare Life Sciences).
Microcarrier preparation
In bioreactor cultivation of the Vero cells, the microcarrier concentration was 3 g/L. Cytodex 1 microcarriers (GE Healthcare Life Sciences) were hydrated in PBS in a siliconized glass bottle (Sigmacote™, Sigma-Aldrich Co.) and washed three times with PBS prior to autoclaving for 15 min at 121ÅãC. Before use, the microcarriers were washed with cultivation medium.
Bioreactor cultivation
Cultures at 10 L scale were grown in a WAVE Bioreactor 20/50 system (GE Healthcare Life Sciences). WAVEPOD II controller (GE Healthcare Life Sciences) was used to control temperature, pH, DO, and agitation. The culture parameters were maintained at 37ÅãC, pH 7.1, DO 30%, and the agitation settings were 10 rpm/5Åã. Cultures at 50 L scale were run in a WAVE Bioreactor 200 system (GE Healthcare Life Sciences). Cultivation conditions were the same as for the 10 L cultures except for the agitation setting, which were changed to 6 rpm/5Åã.
Scale-up procedure and bead to bead transfer
Inoculum grown in cell factories was used to seed a 10L microcarrier culture. The 10 L culture was subsequently used to inoculate a 50 L culture by bead to bead transfer. For cell transfer to the 50 L microcarrier culture, the heating and agitation were switched off and the microcarriers were allowed to settle. The cells were washed twice in PBSEDTA inside the Cellbag 50L bioreactor (GE Healthcare Life Sciences). For wash, the supernatant (90% of the volume) was removed and partly replaced with PBS-EDTA. After a second wash, the microcarrier suspension was transferred to a Sigmacote siliconized glass bottle. In a laminar flow hood, the residual buffer was removed and trypsin-containing buffer solution (0.2% trypsin and 0.02% EDTA in PBS w/o Ca2+ , Mg2+ ), corresponding to 250% of the settled carrier volume, was added. The bottle was incubated at 37ÅãC in a water bath and the suspension was mixed every 5 min. After 20 min, cell detachment was verified microscopically and the supernatant transferred to a glass bottle. The microcarriers were washed twice with cultivation medium and the supernatants were pooled. The cell suspension was used to inoculate a 50 L culture in a Cellbag 200L bioreactor (GE Healthcare Life Sciences). 29-0435-48 AB 3
Cell sampling and counting
During bioreactor cultivation, samples were taken daily to determine cell concentration and morphology as well as concentrations of selected metabolites. Microcarrier suspension was withdrawn via the sample port while the base unit was rocking continuously. A 1 mL sample wastransferred to a tube. After settling of the microcarriers, 800 μL supernatant was removed and replaced with an equal volume of 0.1% crystal violet in 0.1 M citric acid and 1% Triton™ X-100. The suspension was vigorously mixed for 45 s and the released nuclei were counted using a hemocytometer. Cell morphology and attachment to microcarriers were determined using an inverted microscope with attached camera (Eclipse TS100, Nikon Instruments Inc., Melville, NY, USA). The concentrations of glucose, lactate, glutamine, glutamate, and ammonium were measured in a Bioprofile Flex™ analyzer (Nova Biomedical Corporation, Waltham, MA, USA).
Virus stock preparation and infection
Influenza A/Solomon Islands/3/2006 (H1N1) IVR145 WHO (egg-derived) was adapted through a number of passages in Vero cells. A working virus bank was established and tested or infectivity by using 50% tissue culture infective dose (TCID50 ). Cells were washed in serum-free medium prior to infection. For washing, the heating and agitation were switched off, the microcarriers were allowed to settle inside the Cellbag bioreactor, and 80% of the supernatant was removed and replaced with serum-free medium. Two washings were performed for efficient removal of serum. The cells were infected during the exponential growth phase at a multiplicity of infection of 4 Å~ 10-3 (the ratio infectious virus particles to number of cells) and a trypsin concentration of 2 mg/L.
Harvest
During virus growth, samples were taken daily to visually inspect the cells for cytopathic effect (CPE) and to determine HA concentrations and virus titers. The cultures were harvested 3 to 4 d after infection. The supernatant was clarified by depth filtration and influenza virus was purified as described in application note 29-0435-49 AA.
Analytical methods
The amount of infectious virus particles was measured by TCID50 assay. This assay measures the dilution of the sample that generates CPE in 50% of the cells. Vero cells were seeded in 96-well microplates and incubated in 5% CO2 at 37ÅãC overnight. The cells were infected with 10-fold serial dilutions of virus sample, followed by incubation for 5 d in 5% CO2 at 33ÅãC. The cells were visually inspected for CPE and the TCID50 titer was calculated according to the method of Reed and Muench (5). The outcome is presented as a log10 titer (10xx TCID50 units/mL). Total amount of virus particles was measured in Virus Counter 2100 (ViroCyt, Denver, CO, USA). The measurement is based on flow cytometry. Proteins and nucleic acids in the sample were labelled with two different flourescent dyes. A simultaneous signal of protein and nucleic acid was counted as an integral virus particle. Virus Counter measures all virus particles present, both infective and non-infective.
HA was quantified in Biacore T200 with sensor Chip CM5 (GE Healthcare Life Sciences). Recombinant HA protein was immobilized on the dextran matrix. Virus-containing samples were mixed with a fixed concentration of antiserum and injected over the chip surface. Free antibodies (not bound to virus at equilibrium) bound to the surface HA. Low concentration of virus in the sample resulted in a high antibody binding to the surface, while a high virus concentration resulted in low antibody binding. A HA calibration curve, based on reference material from the National Institute for Biological Standards and Control (NIBSC), was included three times in each run: at start, in the middle, and at the end. These calibration curves were used for software normalization to obtain accurate concentrations of the samples.
Results and discussion
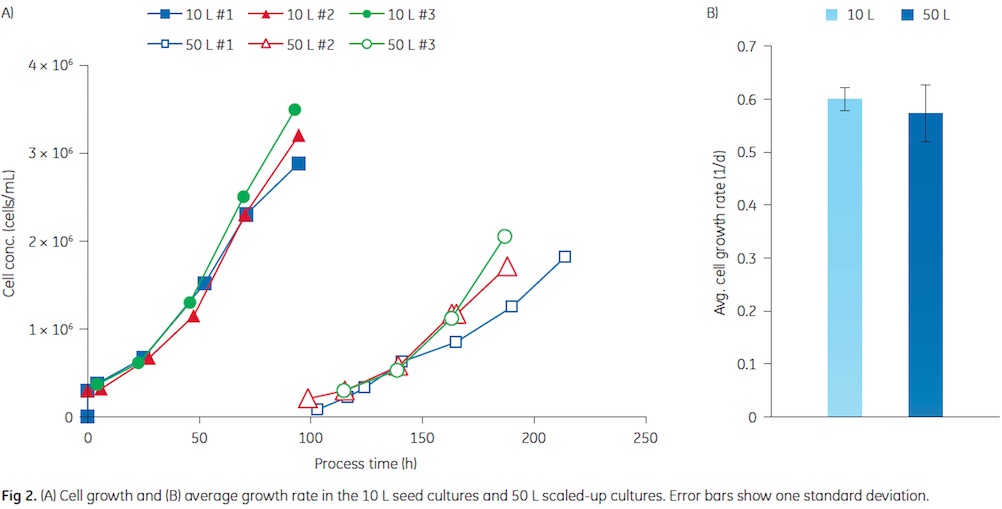
The cell growth in the 10 and 50 L cultures is shown in Figure 2. Similar growth was observed in three consecutive experiments. In the seed cultures, final cell concentrations of about 3 Å~ 106 cells/mL were achieved. Cells from the seed cultures were used to inoculate the 50 L production culture. A split ratio of 1:5 was used throughout this study. In both cultivation volumes, Vero cells attached to the microcarriers and started to grow without a lag phase. In previous experiments, delayed cell growth was observed after the bead to bead transfer (data not shown). Reducing the incubation period with trypsin from 30 to 20 min seemed to overcome this problem. Most probably, this is caused by a reduced effect of the protease on the cell surface receptors, especially integrins, which are required for reattachment and growth of adherent cells (6). As shown in Figure 2, the growth rates in the 10 and 50 L culture were similar. The 50 L cultures were infected with influenza virus at a cell concentration of about 2 Å~ 106 cells/mL that was obtained about 4 d after the bead to bead transfer. The first 50 L culture was extended by one day to reach the desired cell concentration. The reason was probably the lower inoculum concentration compared to the following two runs.
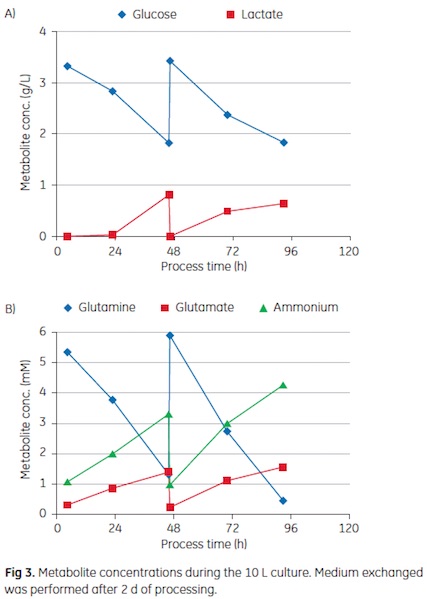
Representative metabolite concentrations during the four-day culture are shown in Figure 3. In both cultivation volumes, an 80% to 90% medium exchange was performed after 2 days of processing to maintain sufficient nutrient concentrations. As shown in the graph, the residual glucose concentration was kept above 1.5 g/L and the lactate concentration was maintained below 1 g/L. As a key energy source, glutamine is a a critical nutrient for cells and heavily consumed during growth. Hence, a medium exchange was required after two days of cultivation in order to prevent limiting concentrations. It is crucial to prevent depletion, as this can trigger apoptosis (7). Medium exchange may be more favorable than plain feeding of glutamine, as the accumulated ammonium concentration already exceeded 4 mM. The washing with serum-free medium before virus infection supplied the cultures with fresh nutrients and removed catabolites. A low ammonium concentration seems to be desirable during virus propagation, as it has been shown that elevated concentrations of this catabolite can prevent infection of the host cell (8).
The scale-up strategy from static cell factories to 10 L seed cultures, and then further expansion to 50 L cultures by bead to bead transfer, was shown to be reproducible. The cells attached and grew on the new microcarriers also after bead to bead transfer. However, it is important to optimize the cell concentration at bead to bead transfer. The optimal cell concentration is dependent on the cell line and cultivation conditions. The morphology of Vero cells at different stages of the process is shown in Figure 4. The cells spread out and reached confluence on the microcarriers. CPE was observed 2 to 3 days after virus infection.
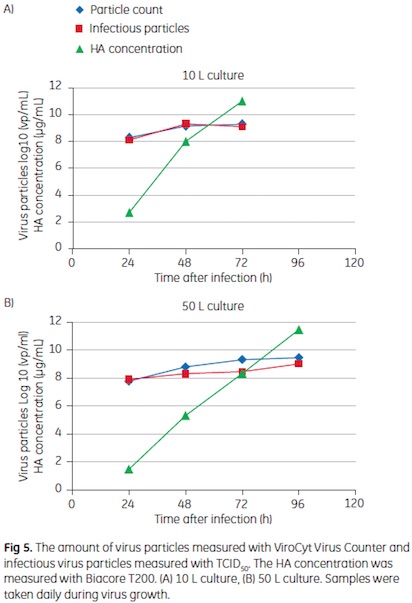
In Figure 5, a comparison between virus growth in a 10 L and a 50 L culture is shown. The smaller volume was inoculated directly from Cell Factory, the larger volume via bead to bead transfer as described above. Both cultures were infected at similar cell concentrations of approximately 2 Å~ 106 cells/mL. The amount of infectious virus particles, as determined by a TCID50 assay, as well as the total virus amount, according to virus count measurements, increased with cultivation time. The majority of virus particles were formed during the first two days after infection. The HA concentration increased continuously throughout the
infection phase and peaked at the time of harvest. While a slightly slower virus growth was observed in the 50 L culture, the same final concentrations were obtained. At harvest, the infectious virus concentration in both cultures was 109 TCID50 units/mL. The total amount of virus particles showed a good correlation to the TCID50 measurements. Interestingly there was no significant difference in virus count between the ViroCyt system and the TCID50 assay. The HA concentration at harvest was about 12 .g/mL in both cultivation volumes.
Conclusions
This case study shows a robust and reproducible scale-up procedure demonstrated by three consecutive cultivation experiments, in which TCID50 values of 109 infectious virus particles/mL, was achieved. Assuming a recovery of 25% for the overall process and a dose requirement of 107 TCID50, more than 1.5 million doses of monovalent live attenuated influenza vaccine could be produced from a 50 L cell culture. The processes were run in single-use WAVE Bioreactor 20/50 and WAVE Bioreactor 200 systems. Compared to virus production in stainless steel bioreactors, the setup and turnaround time can be substantially reduced by the use of disposable systems that make time-consuming cleaning and sanitization procedures redundant. The described process is a fast and convenient alternative to more traditional methods. The case study described in this application note is not a fully optimized process. Further optimization of the process is necessary prior to use in vaccine manufacturing.
References
1. Faster scale-up of a two-step Mab process with AxiChrom™ columns and ReadyToProcess solutions. GE Healthcare, Upstream and Downstream, 28-9863-33, Edition AA, 3:14-16 (2010)
2. Liu C.C., Lian W.C., Butler M., Wu S.C. High immunogenic enterovirus 71 strain and its production using serum-free microcarrier Vero cell culture. Vaccine 25:19-24 (2007)
3. Rourou S., van der Ark A., van der Velden T., Kallel H.. A microcarrier cell culture process for propagating rabies virus in Vero cells grown in a stirred bioreactor under fully animal component free conditions. Vaccine 25:3879-3889 (2007)
4. Kistner O., Howard M.K., Spruth M., Wodal W., Bruhl P., Gerencer M., Crowe B.A., Savidis-Dacho H., Livey I., Reiter M., and others. Cell culture (Vero) derived whole virus (H5N1) vaccine based on wild-type virus strain induces cross-protective immune responses. Vaccine 25:6028-6036 (2007)
5. Reed L.J. and Muench H. A simple method of estimating fifty per cent endpoints. American Journal of Epidemiology 27:493-497 (1938)
6. Brown M.A., Wallace C.S., Anamelechi C.C., Clermont E., Reichert W.M., Truskey G.A. The use of mild trypsinization conditions in the detachment of endothelial cells to promote subsequent endothelialization on synthetic surfaces. Biomaterials 28:3928-3935 (2007)
7. Mendonca R.Z., Pereira C.A. Cell metabolism and medium perfusion in VERO cell cultures on microcarriers in a bioreactor. Bioprocess Engineering 18:213-218 (1998)
8. Genzel Y., Behrendt I., K.nig S., Sann H., Reichl U. Metabolism of MDCK cells during cell growth and influenza virus production in large-scale microcarrier culture. Vaccine 22:2202-2208 (2004)
GE and GE monogram are trademarks of General Electric Company.
AxiChrom, .KTA, Biacore, BioProcess, Cellbag, Cytodex, ReadyCircuit, ReadyToProcess, ULTA, and WAVE Bioreactor are trademarks of General Electric Company or one of its subsidiaries. Accutase is a trademark of Innovative Cell Technologies Inc. Bioprofile Flex is a trademark of Nova Biomedical Corporation. Nunc and Cell Factory are trademarks of Thermo Fisher Scientific Inc. Pluronic is a trademark of BASF. Sigmacote is a trademark of Sigma-Aldrich Co. Triton is a trademark of Union Carbide Chemicals and Plastic Company Inc.
All other third party trademarks are the property of their respective owner. ˝ 2013–2014 General Electric Company—All rights reserved. First published Apr. 2013 All goods and services are sold subject to the terms and conditions of sale of the company within GE Healthcare which supplies them. A copy of these terms and conditions is available on request. Contact your local GE Healthcare representative for the most current information.
GE Healthcare UK Limited
Amersham Place, Little Chalfont
Buckinghamshire, HP7 9NA
UK
GE Healthcare Europe, GmbH
Munzinger Strasse 5
D-79111 Freiburg
Germany
GE Healthcare Bio-Sciences Corp.
800 Centennial Avenue, P.O. Box 1327
Piscataway, NJ 08855-1327
USA
GE Healthcare Japan Corporation
Sanken Bldg., 3-25-1, Hyakunincho
Shinjuku-ku, Tokyo 169-0073
Japan
29-0435-48 AB 04/2014
For local office contact information, visit
www.gelifesciences.com/contact
www.gelifesciences.com/bioprocess
GE Healthcare Bio-Sciences AB
Bj.rkgatan 30
751 84 Uppsala
Related Topics and Keywords
cell culture-based vaccine production Single-use bioprocessing equipment. Single-use Tecnooogies disposable bioreactor systems microcarriers Microcarrier preparation Bioreactor cultivation bead to bea, manufacturing, Single-use technologies
Subscribe to our FREE newsletter and WEBINAR UPDATES
We will not sell or give your information to a third party. See our Privacy Policy