Neusilin®-Polymer Extrudates for the Development of Amorphous Solid Dispersions
Posted on March 26, 2014
ABSTRACT
The aim of this study was to develop and characterise indomethacin (INM) based solid dispersions. Neusilin® (NEU)-polymer blends were used as a suitable carrier via hot melt extrusion (HME) processing for the formation of stable amorphous solid dispersions of indomethacin.
INTRODUCTION
Solid dispersions are similar to a group of solid products consisting of at least two different components; generally a water soluble matrix and a water insoluble drug. However, the matrix can either be crystalline or amorphous. HME has been used to prepare solid dispersion of various APIs, where polymers or lipids are used as carriers [1]. Further investigations relating to the use of Neusilin® (an inorganic excipient) as a co-processing agent in HME are of significant interest in the development of solid dispersions. Neusilin® is a synthetic magnesium aluminometasilicate (MAS) and shows exceptional excipient properties for improved API delivery of pharmaceutical preparations. Here, we report the use of Neusilin® as a co-processing excipient with cationic methacrylate hydrophilic polymer (MEPOL) for the formation of a solid dispersion of INM and the solid state characteristics of the API. A possible intermolecular interaction has also been evaluated in the foregoing study.
MATERIALS AND METHODS
Indomethacin formulations with NEU/MEPOL (20-40% drug w/w) were well mixed in 100 g batches for 10 min each prior to extrusion. A Turbula TF2 Mixer was used to blend the powder formulations thoroughly. Extrusion of all formulations were performed using a Eurolab 16 twin screw extruder (Thermo Fisher, Germany) equipped with a 2 mm rod die with a screw speed of 50 rpm (feed rate 0.6kg/hr). The temperature profile used for all formulations was 50oC/100°C/120°C/135°C-135oC/135°C (from feeding zone → die). The produced extrudates (strands) were cut into 1 mm pellets by using a pelletizer (ThermoFisher, Germany). The pellets were micronised by a Rotor milling system to collects the granules below 250 µm threshold (Retsch, Germany). The extrudates were characterized via differential scanning calorimetry (DSC), XRPD, SEM, FT-IR and in vitro dissolution studies.

Figure 1 – SEM images of INM/NEU/MEPOL extrudates.
RESULTS AND DISCUSSION
The SEM photographs (Fig.1) revealed no INM crystals on the surface of the extrudates. DSC thermograms of the physical mixtures of the INM/NEU/MEPOL (20-40%) showed two endothermic thermal transitions (Fig. 2) corresponding to the Tg of the polymer (~59oC) and the melting peak of the drug at relatively higher temperature (166-187oC). None of these transitions are detected in extruded formulations apart from a Tg shifted towards a lower temperature in all extrudates indicating the presence of molecularly dispersed INM (Tg = 42oC) in the NEU/MEPOL matrices.
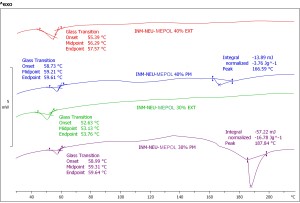
Figure 2 -DSC thermograms of INM/NEU/MEPOL solid dispersions.
The XRPD analysis (Fig. 3) showed no distinct peaks representing the crystalline state in any of the extruded formulations, while INM occurs in the crystalline form in all physical mixtures.
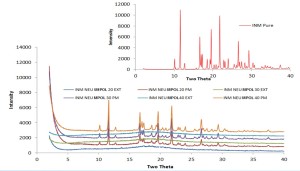
Figure 3 – XRD patterns in powder X-ray Diffraction.

Figure 4 – (i-ii) FT-IR spectra of INM/NEU/MEPOL extrudates and pure subtances.
Indomethacin exists as dimers in the crystalline state due to benzoyl, carbonyl and carboxylic acid containing structure [2]. The FTIR spectra of the crystalline state of INM showed the acid dimer peak at 1715 and at 1690 cm-1, respectively (Fig. 4). The absorbance of the drug dimer peaks start getting broader and shift to 1745 cm-1 with the increase of INM loadings, indicating the amorphicity of INM in the extruded formulations. Furthermore, the a new absorbance peak at 2950 cm-1 in extrudates indicates a possible intermolecular interaction between the MEPOL and the drug. Further investigations using XPS and/or EDX could provide further evidence of such interactions.
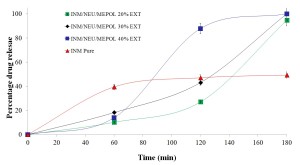
Figure 5 – In vitro dissolution studies of INM based extruded formulations.
As expected, the extrudates with 20-30% INM loadings showed faster release of INM (80% in 180 min at pH 6.8) compared to that of the physical mixtures at all drug/polymer ratios (data not shown) and the pure INM itself (Fig.5).
CONCLUSIONS
When extruded with NEU-MEPOL, at different drug loadings, INM occurred in the amorphous state. The FTIR studies provided evidence that MEPOL interacts strongly with INM to form solid molecular dispersions.
To learn more please visit us at http://www.fujichemical.co.jp/english and http://www.neusilin.com/.
REFERENCES
[1] Maniruzzaman M, Rana MM, Boateng JS, Mitchell JC, Douroumis D. Drug Dev Ind Pharm 2013, 39(2):218-27.
[2] Gupta MK, Vanwert A, Bogner RH.. J Pharm Sci 2003; 92 (3): 536-51.
Related Topics and Keywords
amorphous, BCS class II, BCS class IV, Crystalline, dry granulation, Excipient, excipients, HME, Hot melt extrusion, hydrophilic, inorganic excipient, insoluble drugs, manufacturing, polymers, poorly water soluble drugs, solid dispersion, Spray-drying
Subscribe to our FREE newsletter and WEBINAR UPDATES
We will not sell or give your information to a third party. See our Privacy Policy

















