Performance Comparison Of CHO-S CellCultivation and mAb Production in the Allegro™ STR 200 Bioreactor and Conventional Single-Use Bioreactor
Posted on November 12, 2019
Pall Biotech
Introduction
The benefits of single-use technologies for upstream use now are much more prevalent and heavily adopted within biopharmaceutical industry, causing an increase demand for single-use stirred tank bioreactors to evolve toward a more robust design that has simplified operation and set up to end users, while safe guarding the product. To respond to that demand, Pall has developed the Allegro STR single-use stirred bioreactor family (Figure 1).
Currently available in 4 sizes, Allegro STR 50, 200, 1000, and 2000 L bioreactors. This product line has innovative features that differentiate it from conventional cylindrical single-use bioreactors (SUB) available on the market. For instance, its direct-bottom mechanically driven impeller allows large range of power inputs from 0.002 W/kg up to 0.3 W/kg, while the macro sparger seated below the three 45 degree elephant ear blades results in high oxygen transfer rates (kLa up to 40h-1) and short mixing time (minimum t ~10 s). Its novel cubical like shape design integrates 3 baffles into the stainless tank walls, which alleviates swirling and vortex formation, a common drawback of a cylindrical SUB. Moreover, its large accessible front door and the semi-automatic biocontainer inflation greatly eases biocontainer installation and reduces minimum operator contact with Allegro plastic film and risk to damage it.
Besides state-of-the-art design and usability features, scalability performance of Allegro STR single-use stirred bioreactor family has been demonstrated. More information are available in application notes USD29801, USD31362, USD31803, USD29264, and USD31355 describing respectively engineering performance of the Allegro STR 200, 1000, and 2000 bioreactor and CHO cell culture performance in the Allegro STR 200, 1000 bioreactors. These studies highlighted that the critical parameters to be kept constant from one scale to another are the power input per volume (P/V) for agitation and the superficial gas velocity (vs) for aeration. This will successfully scale up a process at constant kLa, keeping equivalent mixing time and shear stress environment in the bioreactor.
This application note will demonstrate comparative cell culture performance in the Allegro STR 200 bioreactor compared to a conventional cylindrical shaped single-use bioreactor. A fed-batch cultivation of a CHO cell line expressing a recombinant monoclonal antibody, in a chemically defined, animal-derived component-free (ADCF) medium was performed. Both bioreactors operate at the same power input per unit volume (P/V=50 W/m³) and same constant air flow rate is added through the sparger to strip equally the dissolved CO2 (0.02 vvm). Level of Dissolved Oxygen (DO) is automatically kept at 40% +/- 10% by addition of air and O2 via a PID based control loop. pH is maintained at 7.2 +/- 0.2 by automatic addition of CO2 in the sparger or 1M sodium hydroxide solution via a double-sided PID based control loop.
Materials and Methods
Bioreactors
• Allegro STR 200 single-use bioreactor system with heat mat (Pall)
• Allegro STR 200 single-use biocontainer (Pall)
• Cylindrical single-use bioreactor with two pitched blade impellers
• Flexsafe♦ RM wave bioreactor (Sartorius)
• 125 and 500 mL Erlenmeyer shake flasks (Corning♦)
Analytics
• Polarographic oxygen sensor InPro 6800 (Mettler Toledo♦) – Allegro STR 200 bioreactor
• pH sensor (EasyFerm Hamilton) – Allegro STR 200 bioreactor
• PreSens single-use pH and DO sensors – Conventional bioreactor
• BioProfile 100plus ID 2 (Nova Biomedical)
• Cedex HiRes and Cedex Bio Analyzer (Roche)
• Shimadzu Prominence HPLC System
• Waters Xevo G2 LC-MS and Acquity H-Class Bio UPLC
Cell Line, Medium and Supplements
• CHO-S Clone 38 cell line, producing a human IgG antibody
• ActiPro♦ Medium (GE, HyClone)
• 4 mM Ultraglutamin 1 (Lonza)
• 1x HT Supplement (Life Technologies)
• Puromycin (1.25 g/L, 10 mg/mL)
• Cell boost♦ 7a and Cell boost 7b (GE, HyClone)
• Glucose solution (45%, 450 g/L)
• Antifoam C (Sigma Aldrich) diluted in H2O (1:1000) end concentration 1 ppm
Inoculum Preparation
For this experiment, a cryovial containing CHO-S-38 cell line expressing a humanized IgG1 monoclonal antibody that binds HER2 was thawed in a 125 mL Erlenmeyer shake flask with 30 mL working volume. Passage was performed when the cell count was at or approaching the mid-exponential point of the log phase growth curve. Cells were maintained in 500 mL Erlenmeyer shake flasks with a working volume of 100 mL, using a seeding density of 0.4 x 106 cells.mL-1 in Hyclone ActiPro medium, containing 4 mM Ultraglutamine, 1xHT-Supplement and 1.25 mL.L-1 Puromycin (10 mg.mL-1). The cultivation parameters were set as followed: 37 °C, 80% rel. Humidity, 8.0% CO2, 108 rpm and 25 mm shaking diameter.
Volume expansion for inoculum production was performed in a 50 L Flexsafe RM wave bioreactor (Sartorius Stedim Biotech). Inoculation density was 0.4 x 106 cells.mL-1 while in total three 500 mL shake flasks, and were pooled together with 820 mL of cell suspension used for inoculation with a starting working volume of 12.5 L. On day two and three, a feeding of 6.25 L supplemented ActiPro medium containing 1.25 mL.L-1 puromycin was performed. The wave bioreactor was operated in a similar fashion to shake flask operation at 37 °C, pH 7 +/- 0.2, DO 40% +/- 10% controlled with O2 and 0.07 vvm overlay flow (0.875 lpm). Due to volume addition, alterations were done on aeration to keep gas hold-up (vvm) constant and on agitation from 20 rpm/7° angle at start, to 30 rpm/8° 48 h.p.i and 35 rpm/8° 72 h.p.i. The cell density reached 7.6 x106 cells.mL-1, after four days of cultivation to inoculate respectively the Allegro STR 200 bioreactor and the single-use conventional stirred tank bioreactor at a seed density of 0.4 x 106 cells.mL-1.
Bioreactor Set-Up
The engineering performance technology transfer modelling to yield biochemical parameters for this comparison study were determined based on previous studies. Therefore, sparger air gas hold up (VVM, volume flow per unit of liquid volume per minute) and power input per volume (P/V) were kept constant in both systems (Table 1). Starting working volume was set up at 160 L in order to fully cover impellers of the conventional single-use bioreactor to maximize mixing efficiency that is compared in both systems. The pH was adjusted using sparging CO2 and 1 M sodium hydroxide. The DO was controlled via sparger with O2.
A day prior to inoculation, the biocontainer was inflated by adding air via the 0.2 μm absolute filter of the overlay gas line. The pH and DO sensors were calibrated and sterilized by autoclaving at 121 °C for 15 minutes, before being inserted into the single-use bioreactor biocontainer via the Kleenpak™ sterile connectors. 150 L of ActiPro medium containing 4 mM Ultraglutamine (Lonza) and 1 x HT-Supplement (Gibco) was pumped in via the line at the top of the bag through one of the addition lines. Agitation was set at 73 rpm and overlay airflow at 5 L/min with no sparging. The temperature probe resistance temperature detector (RTD) was inserted into the single-use bioreactor biocontainer temperature sleeve before turning on the heater mat to allow the liquid to reach the set point of 37 ºC overnight to equilibrate. The same set up was performed on both systems apart from pH and DO single-use sensors used in the single-use conventional bioreactor, as opposed to traditional inserted pH and DO technology.
Inoculation
Prior to inoculation, the medium was equilibrated to the starting control conditions. Once the pH and DO setpoints were reached, the harvest (transfer out) from the wave bag was directly added into the conventional single-use bioreactor or the Allegro STR 200 single-use biocontainer, at a seeding density of 0.4 x 106 cells.mL-1 and viability greater than 95%. 8.1 L of inoculum has been added in the conventional single-use cylindrical biocontainer while 7.9 L of inoculum has been added in the Allegro STR 200 single-use biocontainer, bringing the total volume to approximately 160 L.
Fed-Batch Process
The comparison study was based on the operating parameters to ensure a constant power input per unit volume (W.m-3) and constant sparger air gas hold up (vvm). The initial starting volume was approximately 160 L and the feed strategy was initiated 3 days after inoculation to reach 200 L final. It consisted of a daily addition of Cell boost 7a at 2% of culture volume and Cell boost 7b at 0.2% of culture volume (Table 2). Both single-use bioreactor cultures were sampled daily and glucose concentrations were maintained at 4 g.L-1 by regular glucose additions to the culture (Sigma Aldrich, 45% in H20). The generation of foam was reduced by adding antifoam C when required. The study was terminated after 10 days.
Analytics
Culture samples were taken daily to monitor cell density and viability, metabolites, mAb titre and quality analysis such as charge variants, aggregates and glycans profiles. Nutrients and metabolites were measured using the BioProfile 100plus ID 2 (Nova Biomedical). Viable cell density was determined using standard measurement for CHO Cells of a Cedex Hires (Roche) cell counting unit. A CedexBio Analyzer (Roche) was used to measure IgG concentration using the standard kit for human IgG (Roche). The in-line pH sensor was checked for drift with offline syringe samples and adjusted if required. Product quantity was determined by protein-A using the Shimadzu prominence HPLC system. N-linked glycan profiling will be determined by Hillic chromatography (H-Class Bio Acquity UPLC, Waters, Milford, USA) with detection by fluorescence (FLR) Quadropole time-of-flight mass spectrometer (Waters).
Results and Discussion
Comparison of Batch Culture Cell Growth
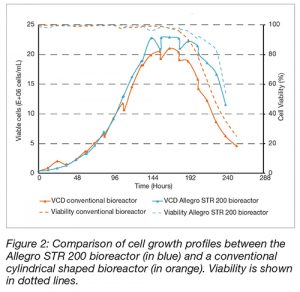
Viable cell densities exhibit culture phase consistency in both bioreactors performed in this experimental work (Figure 2).
In the Allegro STR bioreactor, the cells followed an exponential growth phase to a peak viable cell concentration of 2.29 x 107 cells.mL-1; compare to 2.10 x 107 cells.mL-1 in average the conventional single-use bioreactor (Table 3). The exponential growth phase is highly comparable as demonstrated by the overlapping growth profiles. A specific growth rate of 0.030 hr-1 (doubling time of approximately 23 hours) was observed in the Allegro STR 200 bioreactor culture compare to a specific growth rate of 0.029 hr-1 (doubling time of approximately 24.4 hours).
Comparison of Cell Metabolism
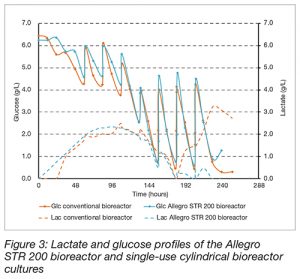
The lactate profiles show a very similar trend except an unexpected lactate production at the end of the culture in the conventional bioreactor (Figure 3). Lactate accumulates for the first 4-5 days, peaking at 2.1 – 2.5 g.L-1, before a shift in cell metabolism occurs whereby it was rapidly consumed. The production and peak lactate levels are seen during the exponential growth phase. Lactate levels then remained low for the remainder of the culture.
Glucose consumption was very similar between both bioreactor types suggesting no influence of the squared shape of the bioreactor on cell metabolism.
mAb Titre Comparison
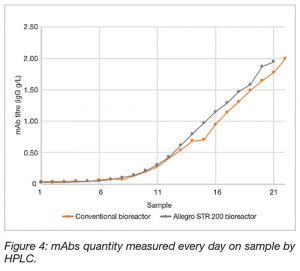
Figure 4 shows the mAb titre in both bioreactors throughout the culture.
In the Allegro STR 200 bioreactor, the antibody concentration at day 10 was 1.94 g.L-1 while in the single-use conventional bioreactor, it increased to 1.78 g.L-1, which is 8% lower (Table 4). Extra analysis (data not shown) using Cedex Bio showed same trend, confirming that the cells in the Allegro STR 200 bioreactor produced higher mAbs levels than the cells in the conventional single-use bioreactor.
Product Quality Comparison
Process conditions of bioreactors can impact the quality of the therapeutic protein produced. The end point corresponding to day 10 of both cultures have been chosen for analysis. Figure 5 shows the main glycan composition of harvest mAb. A similar glycan finger print for both bioreactors was observed (G01, G1F, G2F). Predominantly fucosylated species with the main peak being G0F are produced, as is normal for this IgG antibody.
The amount of aggregates was very similar between both systems with less than 1% difference (Figure 6) and the conventional bioreactor shows a small increase in low molecular weight species (LMW). Overall, the Allegro STR 200 bioreactor processes a high-quality product.
Conclusion
This comparative study demonstrates similar CHO growth, viability and monoclonal antibody quantity and quality whether this process is performed in conventional cylindrical shape bioreactor or Pall’s Allegro STR 200 single-use bioreactor operated at same power input per unit volume, P/V, and same air sparge gas hold up, vvm. It thus confirmed the excellent by design performance and good characterization of Pall’s Allegro STR bioreactors family which help to perform effortless tech transfer based on global and easily available parameters.
Note: Two batches performed in the Allegro STR bioreactor and two batches performed in the conventional single-use bioreactor. One of the two batches for the Allegro STR bioreactor not included in the results due to software error during set-up. One of the two batches in the conventional single-use bioreactor not included in the results due to contamination during sampling. Results therefore based on one batch per bioreactor. Further results may differ.
N=1.
Reference
1. Application Note USD2980: Characterization and Engineering Performance of Allegro™ STR 200 Single-Use Stirred Tank Bioreactor System
2. Application Note USD3136: Characterization and Engineering Performance of the Allegro™ STR 1000 Single-Use Stirred Tank Bioreactor
3. Application Note USD3180: Characterization and Engineering Performance of the Allegro™ STR 2000 Single-Use Stirred Tank Bioreactor4. Application Note USD2926: Cultivation of CHO Cells in Allegro™ STR 200 Single-Use Stirred Tank Bioreactor.
5. Application Note USD3135: Cultivation of Chinese Hamster Ovary (CHO) Cells in Allegro™ STR 1000 Single-Use Stirred Tank Bioreactor System
♦ ActiPro is a trademark of GE Healthcare, Corning is a trademark of Corning Inc., Flexsafe is a trademark of Sartorius Stedim Biotech GmbH gmbh & co, Mettler Toledo is a trademark of Mettler-Toledo.
Contact us at www.pall.com/contact
Subscribe to our FREE newsletter and WEBINAR UPDATES
We will not sell or give your information to a third party. See our Privacy Policy