Good Vibrations: Near-Infrared Spectroscopy in Pharmaceutical Analysis
Posted on September 2, 2015
By: Alfred Steinbach and Stephanie Kappes
Metrohm
Near-infrared spectroscopy (NIRS) is based on the absorption of radiation by matter. Not only does the versatile technique allow the simultaneous identification of substances (active agents, excipients, contaminants), it is also suited to monitor processes such as blending, granulation, and drying.
NIRS – interaction of light and matter
Molecular vibrations are induced in the near-infrared region of the magnetic spectrum (800–2500 nm) – i.e., from the end of the visible to the mid-infrared (MIR) range. The main absorption bands of the functional groups of chemical substances are located in the MIR range and are very strong. The absorption bands of the harmonics and the combination of the fundamental molecular vibrations, however, are in the NIR spectral region. They are significantly weaker and enable direct measurement without sample preparation, while at the same time offering deep insights into the chemical and physical properties of the sample. The strongest overtone absorptions in the NIR range are displayed by compounds with OH, CH, NH, and SH bonds. Because the NIR spectrum represents the result of numerous overlapping absorption bands, it is normally evaluated with multivariate chemometric methods.
Many parameters in a single analysis
NIRS offers numerous advantages over many wet-chemical analytical methods. A diverse range of parameters can be determined simultaneously. NIRS is economical and fast, enabling qualitative and quantitative analyses that are noninvasive and nondestructive.
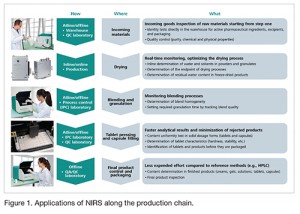
NIRS is an indispensable analysis technique that can be used along the entire production chain – from incoming materials to processing to the quality control of finished products (Figure 1). NIRS meets the requirements of numerous international pharmacopoeias, e.g., USP, Ph. Eur., and JP.
Analyses of diverse matrices
Near-infrared spectroscopy requires no sample preparation and can handle any sample matrix, no matter if it is powders or granulates, tablets or capsules, creams or gels, solutions or suspensions, polymer films, or freeze-dried samples.
Screening through packaged materials
NIRS can even perform determinations on contents sealed in transparent packaging such as glass and films. This is particularly appealing for incoming goods inspections and packaged end products. Handling is so easy that NIRS can be used directly in pharmacies and customs offices.
Nondestructive analysis
NIRS has long been one of the most important and versatile analytical techniques in the pharmaceutical industry – and not just because everybody in the pharma industry is talking about process analytical technologies (PAT) and Quality by Design (QbD). The decisive benefit of NIRS is the possibility of obtaining reliable analysis results in just seconds without altering the material under investigation and without any sample preparation or reagents whatsoever.
PAT and QbD – in search of the best of all methods
Drug manufacturing is subject to strong changes. The FDA’s stated goal is to cut development time for new drugs while at the same time significantly improving quality. This requirement can only be fulfilled with analytical techniques that monitor the entire process – from incoming raw materials to the final inspection. To achieve that, perfect PAT sensors are needed that enable “live” tracking of the manufacturing process. NIRS is the technique that makes this possible. An inline sensor monitors product quality in real time. This prevents out-of-specification products and reduces overall costs.
First example: granulation and drying
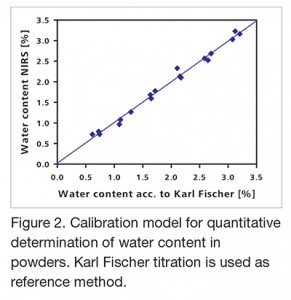
A key manufacturing process in the pharmaceutical industry is the granulation and drying process for powders that precedes tablet manufacturing. This process makes it possible to press powders into tablets in the first place. NIRS is the method of choice for determining the reaction endpoint when pressability is at the optimal point. Probes in the drier or granulator make it possible to track the process in real time. That reduces the process duration and thus increases the drying and granulation capacity of the system. At the same time, it minimizes the deviation from the required set-point values. Figure 2 shows a calibration model for water determination that correlates NIRS to Karl Fischer titration which is the reference method.
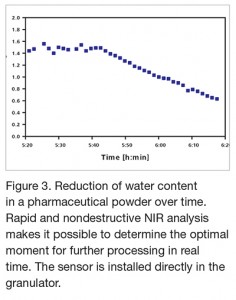
The progressive diminution of the water content during the granulation process, measured by real-time NIRS, can be seen in figure 3.
Second Example: blend homogeneity
Well-mixed active substances for medications are indispensable in the pharmaceutical industry. This applies not only to the pharmaceutical active ingredient but also for lubricants, binders, disintegrants, colorant(s), and antioxidants. Rarely are any of these tested in a normal mixing study. Analysis of the active ingredients is expensive; they are also only rarely analyzed as a rule. The progress of the mixing procedures can be followed conveniently with NIR spectroscopy, using both visual comparisons of data and by means of spectral algorithms. The progress of mixing processes can be predicted in real time with the help of the spectrum when the latter method is used.
Figures 4 through 7 show the earliest, middle, penultimate, and final mixtures of the run, respectively. The correlation improves with each mixing test point. The endpoint of the mixing study must be determined by the analyst as the mixture best correlating with the “standard mixture.”
It is apparent from the data that the use of a spectral matching algorithm can save hundreds of hours in a routine mixing study. For regulatory reasons, however, several samples of the final and penultimate materials must be assayed for content uniformity. Despite these required tests, a significant amount of early testing time may be avoided in future mixing studies.
In accordance with international pharmacopoeias
As a secondary test method, NIRS is recommended in all of the key pharmacopoeias – from the European (Ph. Eur. 2.2.40) to the American (USP<1119>) to the Japanese pharmacopoeia. Metrohm NIRSystems offers instruments that meet the standards for wavelength precision, reproducibility, and photometric noise. Numerous reference standards and user-friendly software make it easy to check the instrument requirements specified in the pharmacopoeias. The pharmaceutical version of the Vision software is fully validated and compliant with 21 CFR Part 11. Metrohm NIRSystems also offers complete IQ/OQ documentation and instrument performance certification (IPC). Documented parameters guarantee that the instrument performs properly.
Subscribe to our FREE newsletter and WEBINAR UPDATES
We will not sell or give your information to a third party. See our Privacy Policy