Oxygen Permeation Rates Through Syringe Components
Posted on November 28, 2016

J.R. Veale and K. G. Victor
Introduction and Background
The recently updated USP 39 <1207> chapter on Package Integrity recommends a move to deterministic analytical methods for assessing container closure integrity of sterile product packages. Here we demonstrate the utility of laser headspace analysis for studying oxygen ingress into syringe packages over a continuum of oxygen ingress rates from permeation to leakage through micron size defects.
All syringes must be a barrier to microbial ingress in order to maintain sterility and some syringes must be a barrier to gas ingress in order to maintain product efficacy (e.g. protect from oxidation or carbon dioxide inducing pH changes).
USP <1207> defines a maximum allowable leakage limit (MALL) as “… the greatest leakage rate (or leak size) tolerable for a given product–package that poses no risk to product safety and no or inconsequential impact on product quality.”
Product sterility and stability requirements have to be understood and a maximum allowable leak limit (MALL) has to be established that will protect the product over the intended shelf life. From a sterility point of view studies have shown that a diffusive leak rate of 10-7 sccs (equivalent to effusive leak rate of 10-6 or 0.2-0.4 micron diameter capillary) is the limit for worst case microbial ingress. From a stability point of view gas ingress rates must be much smaller (e.g 10-10 sccs) in order to keep oxygen below atmospheric levels in a syringe over a 2 year shelf life.
Syringes are more complicated packages than ampoules or vials and have more pathways for gas and microbial ingress. Gas can ingress through and around the plunger or through and around the tip. Further the tip regions are different for luer lock or staked needle configurations. Here we develop Laser Headspace Analysis for studying oxygen ingress through specific parts of a syringe.
Headspace Oxygen Analysis
Oxygen ingress to syringe samples was measured using a Lighthouse Instruments FMS-Oxygen headspace analyzer. The sample holder for the instrument was configured for 1ml syringes. Calibration standards were fabricated from 1ml syringes over the range 0% to 20% and were used to qualify instrument performance. Studies were performed using 1ml staked needle syringes and 1ml luer tip syringes. For all data the measurement uncertainty was equal or better than +/-0.5% absolute.

Syringe Sample Sets
Syringe samples were fabricated from 1ml staked needle and luer tip configurations. Different parts of a syringe were isolated by flame sealing one end of the syringe.

Figure 2. Sample syringes showing how various parts of the syringe can be isolated for study of oxygen ingress. Two types of syringes were studied (staked needle and Luer tip) as well as negative controls (completely flame sealed) and positive controls (flame sealed with micron size laser drilled defects)
Negative controls were flame sealed on both ends. Positive controls were flame sealed on both ends and had 2, 5 or 10 micron laser drilled holes in the syringe body (Lennox Laser). All syringes were flushed with nitrogen before flame sealing. Oxygen ingress was measured over time.
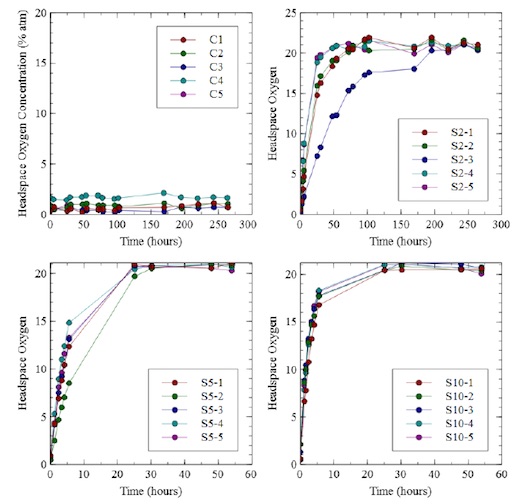
Figure 3. Leak rates through positive and negative control syringes. The negative control samples show no oxygen ingress. Oxygen ingress rates through the positive controls are proportional to the defect diameter.
Syringe Component Results
Figures 4 & 5 present results from measurements of oxygen ingress through isolated regions of both luer tip and staked needle tip syringes. The measurements compare syringe types to negative controls and postive controls with 2 micron laser drilled defects. The oxygen ingress rates are lowest for the staked needle syringes, higher for the luer tip and highest for the syringes with 2 micron defects. In each syringe system the oxygen ingress is dominated by the tip region.

Figure 4. Leak rates through various parts of a luer tip syringe. The negative control samples show no oxygen ingress. The oxygen ingress through the plunger is greater than through th eglass but less than through the tip. The tip region shows the fastest ingress rate.
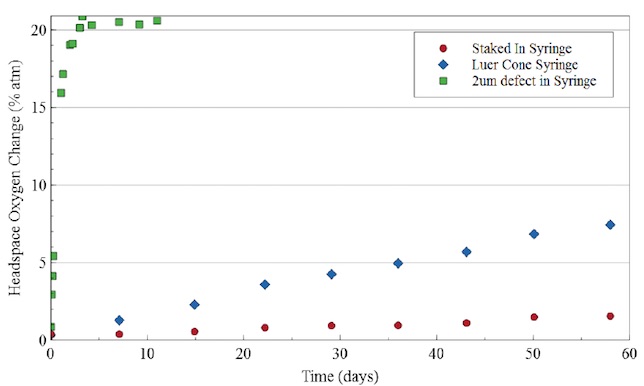
Figure 5. Oxygen ingress rates through unmodified staked needle tip and luer tip syringes compared to a syringe with a 2 micron laser drilled defect. The diffusion rates through the syringe tips are 100-1000x slower than through a 2 micron defect. Diffusive ingress rates are ~10(-6) sccs for the 2 micron defect, 10(-8) sccs for the luer tip and 10(-9) sccs for the staked needle.
Discussion
The data presented in Figures 3-5 show oxygen ingress rates that cover 4 orders of magnitude. A key question is: Do these oxygen ingress rates pose a sterility or stability risk to the product contained in these packages? Figure 6 shows calculations based on a Fick’s Law model of diffusion. The oxygen ingress to a container of volume V can be expressed as:

where D is the diffusion coefficient of oxygen through nitrogen, A0 is the defect area, l is the defect length and V is the container volume. The model predicts the oxygen ingress to each syringe package quite well.
The oxygen ingress rates for both syringe packages fall below the rate considered to be a limit for microbial ingress (0.2-0.4 micron). However the rates are well above the level required to keep oxygen below atmospheric levels over a 2 year shelf life. Care must be taken if oxygen sensitive product is to be packaged in either type of syringe system.
 Figure 6. Calculations of diffusive oxygen ingress to a syringe using a Fick’s Law model. The collected data on Luer tip and staked needle tip syringes fit the data well and allow prediction of oxygen ingress rates over a wide range of defect sizes and/or permeation rates both above and below the microbial limit of 0.2 microns.
Figure 6. Calculations of diffusive oxygen ingress to a syringe using a Fick’s Law model. The collected data on Luer tip and staked needle tip syringes fit the data well and allow prediction of oxygen ingress rates over a wide range of defect sizes and/or permeation rates both above and below the microbial limit of 0.2 microns.
Conclusion
The study presented here allowed for isolated study of oxygen ingress rates through specific regions of a syringe for staked needle and luer tip configurations. The laser headspace method showed an ability to measure diffusive oxygen ingress rates over 4 orders of magnitude from 10-9 sccs to 10-6 sccs. In each syringe system oxygen ingress through the tip region was the dominant ingress pathway and the luer tip configuration showed 10x higher oxygen ingress rates than the staked needle tip. Even though the rates were different in each type of syringe package the overall oxygen ingress rates for both type of package were well below the microbial limit of 0.2 microns but high enough to allow atmospheric levels of oxygen to enter the package over a 12 to 24 month timeframe.
For more information please visit http://www.lighthouseinstruments.com
Related Topics and Keywords
(e.g. protect from oxidation or carbon dioxide inducing pH changes, container closure integrity, continuum, deterministic analytical methods, gas ingress, J.R. Veal, K. G. Victor, laser headspace analysis, leakage, Lighthouse, maintain product efficacy, maintain sterility, microbial ingress, micron size defects, oxygen ingress rates, Oxygen Permeation Rates, Package Integrity, permeation, sterile product packages, studying oxygen ingress, Syringe Components, syringe packages, USP 39 1207 chapter
Subscribe to our FREE newsletter and WEBINAR UPDATES
We will not sell or give your information to a third party. See our Privacy Policy

















